Hydrogels composed of natural biomacromolecules are preferable for 3D cell culture. These biomaterial-based hydrogels are superior to synthetic polymer-based analogs due to not only their innate bioactivity and biocompatibility but also their potential to tune the mechanical and biochemical properties via cross-linking methodologies and tailored molecular weight/sequences. However, most of the commercially available hydrogels with natural biomacromolecule frameworks have some nonideal properties for a 3D cell culture system, such as nontailorable properties, immunogenicity, risks of contamination, inherent batch-to-batch differences, and the poorly defined complex structures. These could interfere with the specific cellular functions of interest. Hence, rational design of 3D cell culture systems with traits of biocompatibility, programmability, easy functionalization, and isothermal casting is highly desired.
In conclusion, they designed and synthesized a dendritic DNA as the building block for self-assembly of hydrogels. The dendritic DNA can gelate at a nanomolar DNA concentration and isothermally within minutes under ambient temperature. In addition, DDH showed a reversible thixotropy, porous morphology, and enzymatic stability conducive to 3D cell culture. With simple handling protocols, multiple cell lines can be cultured repeatedly in DDH with high proliferation and grow into a complex morphology. Bioactive cues can be efficiently incorporated into DDH via multiple strategies to specifically manipulate cell fate. With the easy synthetic and gelation protocol, dendritic DNA shows great potential in encoding versatile stimulus-responsive components toward physical and biological triggers and may overcome the challenges in cost and scale-up for DNA-based hydrogels. DDH could be harnessed as a promising tool in drug release, drug screening, cell therapy, tissue engineering, and other biomedical technologies.
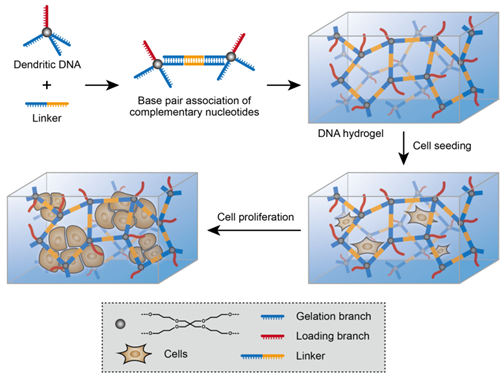
▲Scheme 1. Self-Assembly of the Dendritic DNA Hydrogel (DDH) for 3D Cell Culturea
aDNA branches and segments with the same color are complementary.
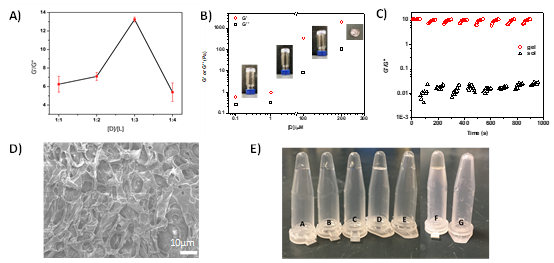
▲Figure 1. DDH characterization.
(A) Optimization of the physical cross-linking among D and L molecules by adjusting the [D]/[L] ratio. (B) Rheological modulus of DDH. G′: storage modulus and G″: loss modulus. Insets: photos of DDH assembled with indicated [D]. (C) Reversible gel-to-sol conversion upon shear strain oscillating from 0.1 to 10,000% ([D] = 200 μM). (D) SEM image of DDH ([D] = 200 μM). (E) Enzyme degradation of 1 μM DDH (A–E) and 1 mM Y-tile DNA hydrogel (F,G) with different amounts of DNase I. (A) 0, (B) 0.25, (C) 0.5, (D) 1.2, (E) 2.5, (F) 0, and (G) 1.2 U per μg of DNA.
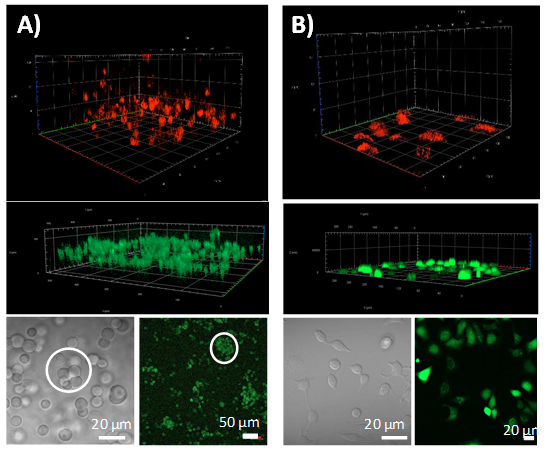
▲Figure 2. A549 cells cultured in C-DDH1
(A) and medium solution (B). 3D stack fluorescence image (top) of cells stained with LysoTracker Deep Red after 48 h of incubation. 3D stacks (middle) and fluorescence images (bottom, right) of cells stained with Calcein-AM (live cells: green) and PI (dead cells: red) to determine cell viability after 96 h. Microscope images (bottom, left) of A549 cells; cell colonies are circled.

▲Figure 3. A549 cells cultured in C-DDH2.
(A) Microscope images and (B) 3D stack fluorescence image of the cell spheroid after 72 h. (C) Cell proliferation in C-DDH2 and 2D solution. (D) Cross-sectional images of the cell spheroid taken at different focus heights. The scale bar is 40 μm. The cells were stained with a CellMask Deep Red plasma membrane.
![▲图4:A549细胞系从C-DDH2中获得细胞后在C-DDH2中进行第二代培养([D] = 200µM)](/sites/default/files/inline-images/6_3.png)
▲Figure 4. Second generation culture of the A549 cell line in C-DDH2 after the cells were harvested from C-DDH2 ([D] = 200 μM).
(A) Microscope images of A549 cells at different times over 8 days. (B) Fluorescence images of A549 cells in C-DDH2 at day 8. Left: confocal image; right: reconstructed 3D image. Cells were stained with Calcein-AM (green: live cells) and PI (red: dead cells) to determine cell viability.
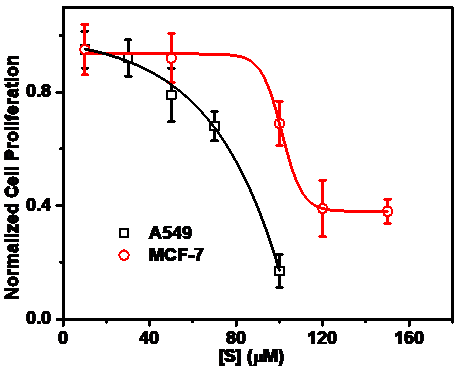
▲Figure 5. Cell proliferation of A549 and MCF-7 cell lines cultured in the aptamer S-loaded hydrogel S-DDH.
![]()
- ▲Abstract:With inherent biocompatibility, biodegradability, and unique programmability, hydrogels with a DNA framework show great potential in three-dimensional (3D) cell culture. Here, a DNA hydrogel was assembled by a dendritic DNA with four branches. The hydrogel showed tunable mechanical strength and reversible thixotropy even under a nanomolar DNA concentration. The cell culture medium can be converted into the hydrogel isothermally at physiological temperature. This DNA hydrogel allows both cancer and somatic cells to be seeded in situ and to achieve high proliferation and viability. The bis-entity of dendritic branches enabled the specific loading of bioactive clues to regulate cell behaviors. Thus, the dendritic DNA-assembled hydrogel could serve as a highly biocompatible, readily functionalizing, and easy-casting gel platform for 3D cell culture.
- ▲Source:https://pubs.acs.org/doi/10.1021/acsami.1c14445
-
▲Research Group of SHAO Fangwei

(Picture, Article: Research Group of SHAO Fangwei; Editor in Charge: ZHANG Yi)





